 
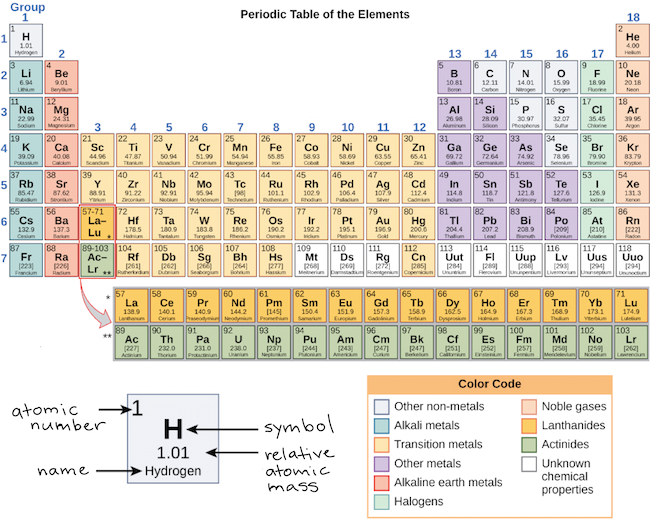
The number of valence electrons for each main group element can be determined by the column, or group, it occupies on the periodic table. Periodic tables always list the atomic number.įigure 2.6.1 - Atomic Number for Each of the Main Group Elements For example, bromine (Br) has 35 protons and 35 total electrons. For neutral atoms, the number of positive protons will equal the total number of negative electrons (zero net charge). The atomic number defines the number of protons in the nucleus of each atom. Neon has 10 electrons - 2 in the first shell, and 8 in the second shell (so eight valence electrons).įigure 2.6.1 below lists the atomic number for the main group elements.Fluorine has 9 electrons - 2 in the first shell, and 7 in the second shell (so seven valence electrons).Oxygen has 8 electrons - 2 in the first shell, and 6 in the second shell (so six valence electrons).Nitrogen has 7 electrons - 2 in the first shell, and 5 in the second shell (so five valence electrons).Carbon has 6 electrons - 2 in the first shell, and 4 in the second shell (so four valence electrons).Boron has 5 electrons - 2 in the first shell, and 3 in the second shell (so three valence electrons).Beryllim has 4 electrons - 2 in the first shell, and 2 in the second shell (so two valence electrons).Lithium has 3 electrons - 2 in the first shell, and 1 in the second shell (so one valence electron).Helium has 2 electrons - both in the first shell (so two valence electrons). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
